Institution: University of Utah
Additional authors:Weiss R, Thompson C, Hussong J, Perkins SL
Session: T Lymphoblastic Leukemia/Lymphoma
HISTORY
51 year-old male with no significant past medical history presented with recent fatigue and weight loss.
DETAILS
A complete blood count demonstrated a white blood cell count of 27.73 x 109/L, hemoglobin 9.7 g/dL, hematocrit 29.7%, and platelets of 11,000 x 106/L. A manual differential count showed 65% atypical immature cells, 30% segmented neutrophils, 3% lymphocytes and 2% monocytes. Peripheral blood smear showed the numerous large atypical cells with scant basophilic cytoplasm, irregular nuclear contours, fine to moderately coarse chromatin and indistinct nucleoli.
A bone marrow biopsy was markedly hyper-cellular (>95%) with near total replacement of the normal hematopoietic elements by sheets of monotonous immature cells. EBER (EBV) in situ hybridization was negative.IMMUNOHISTOCHEMISTRY AND FLOW CYTOMETRY
Flow cytometry performed on a bone marrow core biopsy specimen revealed 95% of the leukocytes to be atypical cells expressing dim CD45, CD34 (partial), CD7, cytoplasmic CD3, CD56 and weak CD33 but negative for surface CD3, CD4, CD1a, CD5, CD8, TdT, CD13, C14, CD15, CD19, CD20, CD61, CD64, CD117, HLA-DR, myeloperoxidase and CD123.
In situ hybridization for EBV (EBER) was negative.CYTOGENETIC FINDINGS
Karyotype analysis showed 46, XY, t(4;8)(q21;q24), add(12)(p11.2), add(14)(q11.2)[cp18]/46,XY[2]. The breakpoint at 8q24 raised concern for a MYC translocation; however FISH with the MYC probe did not show evidence of a rearrangement of this locus. A FISH panel detected loss of 9q34.
MOLECULAR FINDINGS
Molecular analysis was polyclonal for TCR-gamma (TCR-beta not performed).
INTERESTING FEATURES
The differential diagnosis for this case includes NK-cell or T-cell lymphoblastic leukemia/lymphoma, and blastic plasmacytoid dendritic cell neoplasm (PDCN). PDCN was excluded based on lack of skin involvement, clinical features and lack of CD4 or CD123 expression. The distinction of a blastic NK cell versus T-cell lymphoblastic leukemia is further hindered by the fact that there are no routinely used markers that allow for definitive discrimination of NK and T cell precursors. In this case, the polyclonal TCRγ results argue against T-lymphoblastic leukemia/lymphoma. The patient responded transiently to T-ALL therapy but the disease recurred and the patient expired 12 month later. The recognition of this entity is particularly important for development of therapeutic approaches, given the aggressive nature of the disease and lack of response to acute myeloid or acute lymphoid leukemia (Ref: Suzuki eT aL. CD7" and CD56" Myeloid/Natural Killer Cell Precursor Acute Leukemia: A Distinct Hematolymphoid Disease Entity. Blood, Vol 90, No 6 (September 15), 1997: pp 2417-2428).
PROPOSED DIAGNOSIS
WHO provisional entity of NK cell lymphoblastic leukemia/lymphoma.
CONSENSUS GROUP: ADDITIONAL INFORMATION/STUDIES
Additional immunoperoxidase studies performed by the consensus group demonstrated that the neoplastic cells are negative for TIA-1 and granzyme B.
CONSENSUS DIAGNOSIS
T-acute lymphoblastic leukemia/lymphoma, early T-cell precursor immunophenotype with CD56 expression
| Peripheral smear 1000x |  |
| Touch preparation BM 500x | 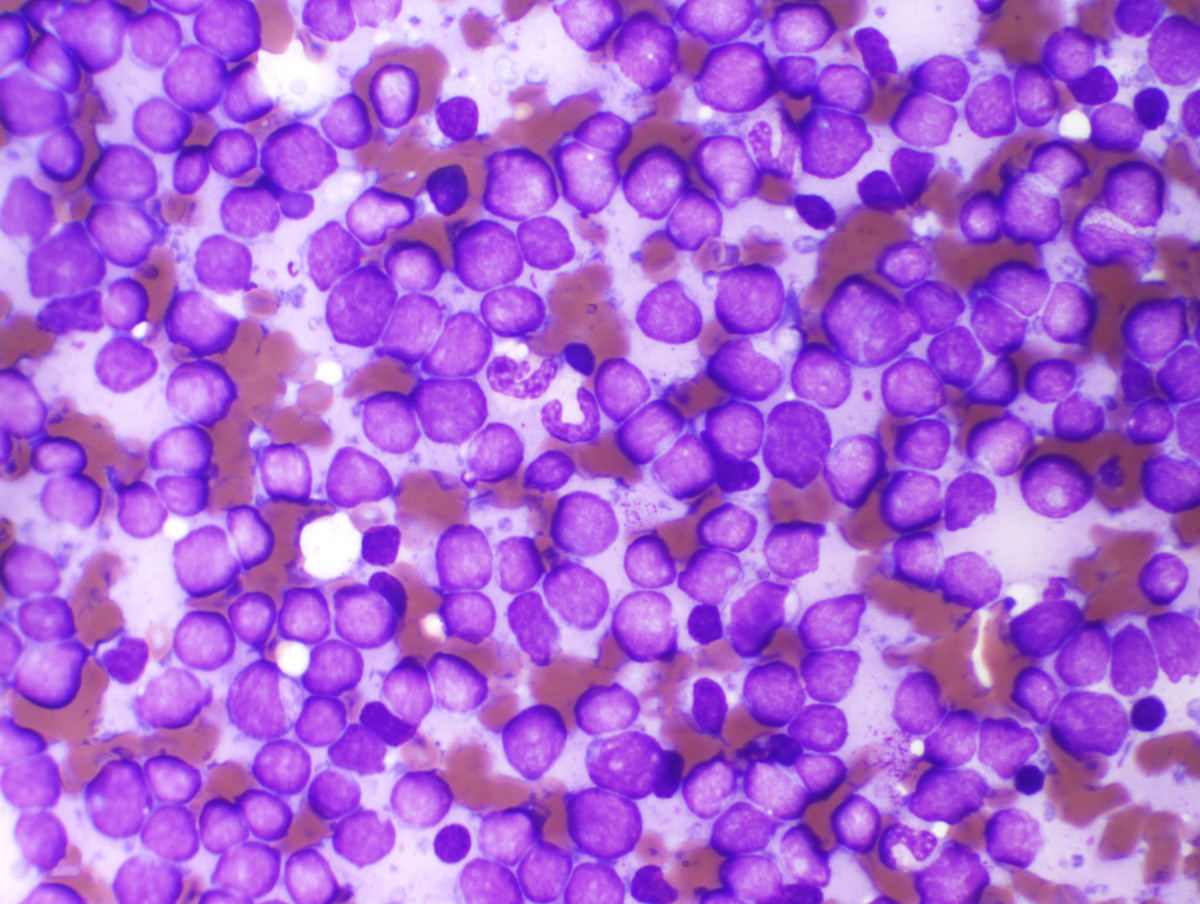 |
| Bone Marrow core 100x | 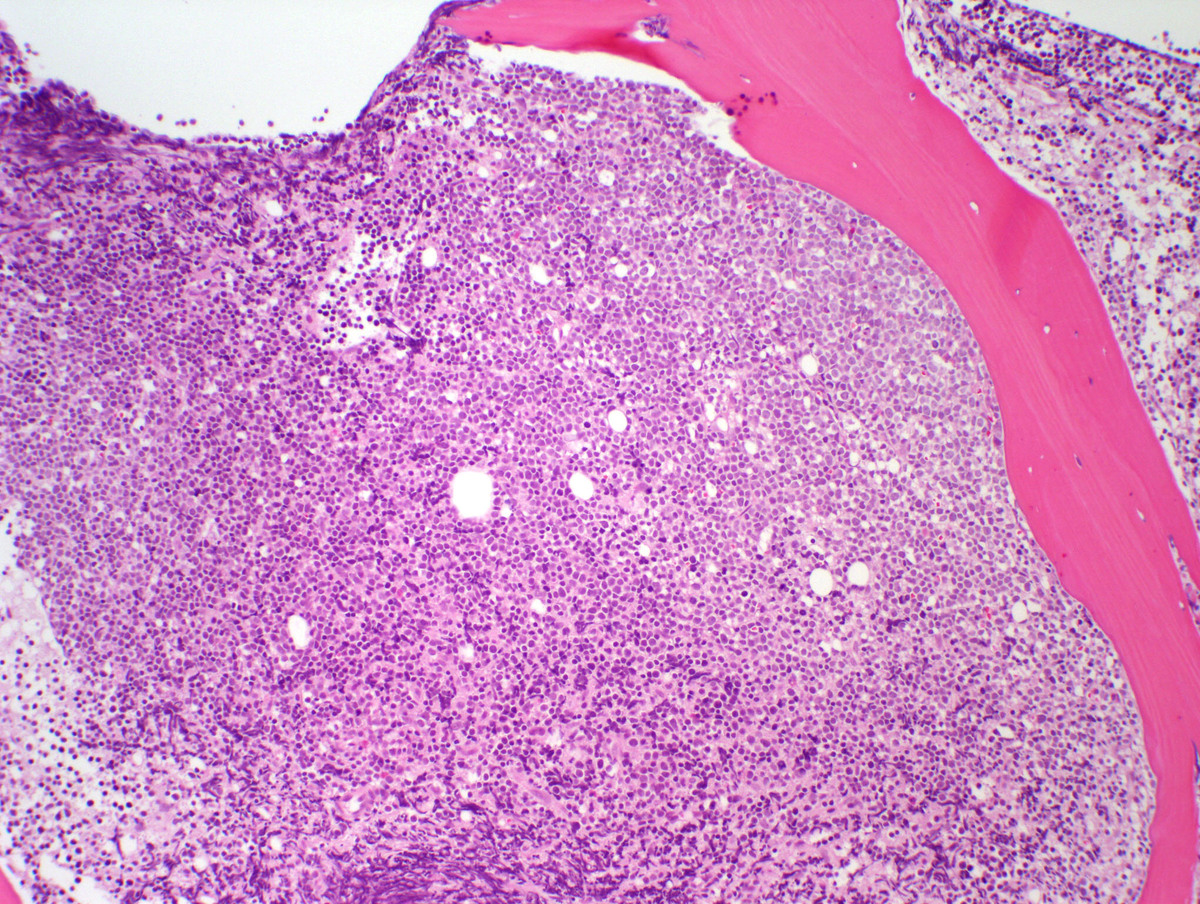 |
| Bone Marrow Core HP 400x | 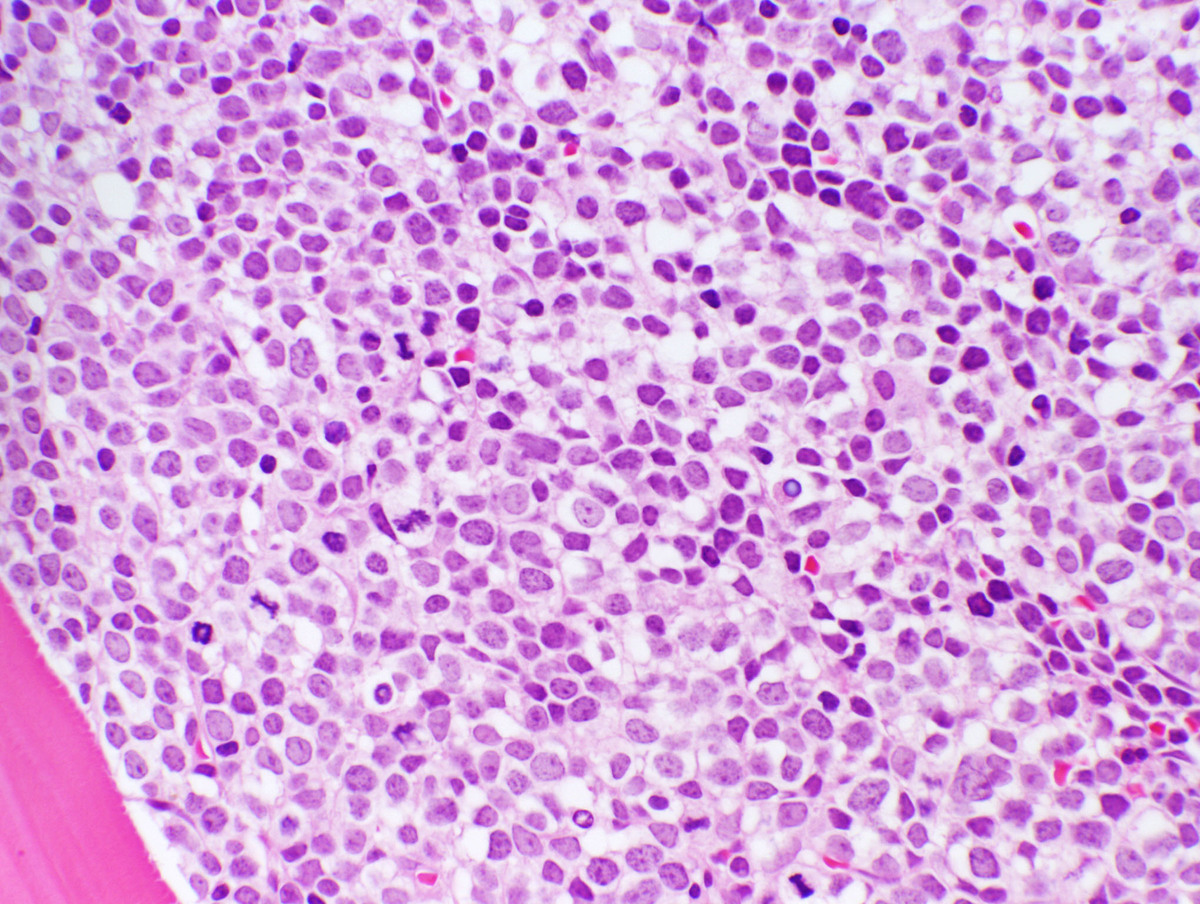 |
| Flow cytometry |  |
| Karyotype | 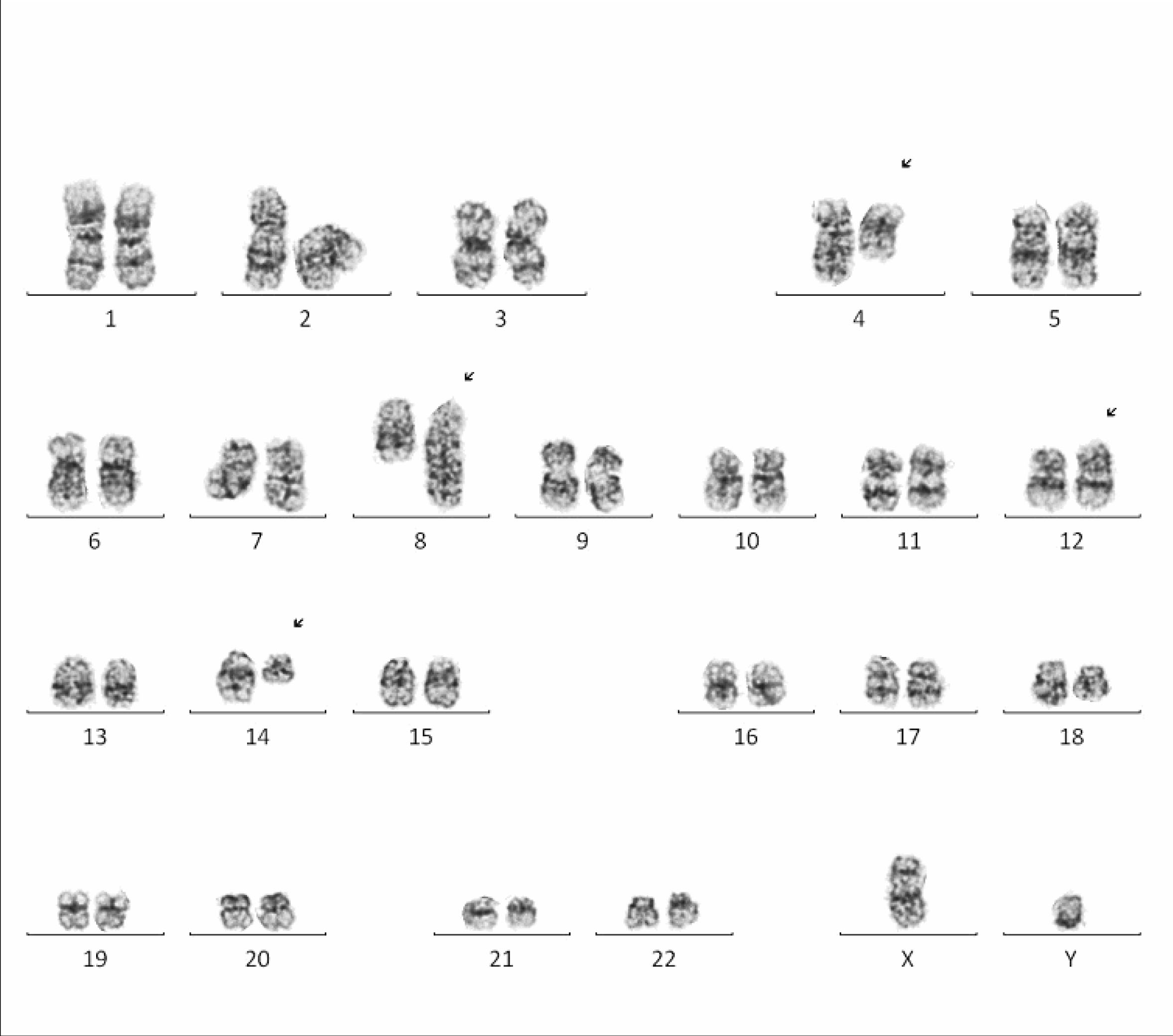 |
| Additional slide 1 - cytoplasmic CD3 (Flow) |  |
| Additional slide 2 - surface CD3 (Flow) |  |
| Additional slide 3 - CD1a (Flow) |  |
| Additional slide 4 - CD1a IHC, 400x |  |