Institution: UT MD Anderson Cancer Center
Session: AML with myelodysplasia-related changes
HISTORY
Patient is a 62 year old female with history of myelodysplastic syndrome (hypoplastic; 10-20% cellularity) with complex karyotype including deletion of 5q, (2004). She received therapy with cyclosporine and prednisone with minor response. She progressed (2006) and was initiated on lenilidomide, achieved a hematological remission and became transfusion-independent through 10/2009. She lost response, was switched to azacitidine (11/2009 - 03/2011), with complete remission and transfusion independency.
In 04/2011, she developed leukocytosis with absolute neutrophilia and left shift with no significant monocytosis. Karyotype was 46,XX,add(7)(p15),del(7)(q22)[17]/90-92,idemX2[cp2]/46,XX[1]. Bone marrow workup demonstrated trilineage dysplasia; 3% blasts, 15% ring sideroblasts. Flow demonstrated 8% blasts. Case was classified as refractory anemia with excess blasts. She presented to UTMDACC (05/2011) with progressive leukocytosis (100K) and thrombocytopenia; was given hydroxyurea. At presentation, Hb 7.6 K, WBC: 55.5 K, platelets 46 K, ANC 36.63 K, absolute monocyte count 10 K. Bone marrow biopsy revealed 90% cellularity and trilineage dysplasia, 6% blasts; case was reclassified as chronic myelomonocytic leukemia. She was put on sapacitabine. In 09/2011, patient developed leukocytosis, absolute monocytosis with blasts in peripheral blood. WBC: 45.4, HGB: 10.9, MCV: 101, Platelet: 55, Neutrophils: 47.0, Lymphs: 3.0, Monos: 23.0, Eos: 1.0, Basos: 14.0, Metas: 4.0, Blasts: 8.0, NRBC: 11,DETAILS
Clot (formalin fixed):
95% cellular; megakaryocyte increased and dysplastic; blasts appear to be increasedAspirate smears: trilineage dysplasia, 52% erythroids; 8% monocytes, 7% blasts patchy, upto 12% focallyButyrate: 11% of total cells positiveIMMUNOHISTOCHEMISTRY AND FLOW CYTOMETRY
CD34+ myeloblasts are 5% with an altered immunophenotype (CD34inc, CD117+, CD13dec, CD33+dec, CD45dec, CD10subset+, CD123dec, CD184dim+, CD5subset+, CD38dec ).
Hematogones are absent.Abnormal patterns or abnormal antigen expression in myelomonocytic cells are immunophenotypically detected. Monocytes are 20% of total cells, CD56+, CD64+, CD14inc, CD123dec, CD184+, HLADRdec.CYTOGENETIC FINDINGS
Karyotype:
46,XX,add(7)(p22),del(7)(q22q34)[20]FISH:monosomy 7 [3.5%]; del(7)(q31) [93%]MOLECULAR FINDINGS
NEGATIVE for mutations in KRAS and NRAS, FLT3 (internal tandem duplication or codon 835/836), KIT (exon 17) , mutation or alteration detected in CEBPA, no mutation in NPM1 (exon 12), IDH1 (exon 4, codons 87 to 138), IDH2 (exon 4), codon 617 of JAK2
Multiplex leukemia translocation assay:NEGATIVE for b3a2, b2a2 and e1a2 transcripts of bcr-abl/ t(9;22)(q34;q11), the short and long forms of PML-RARA/t(15;17)(q22;q21), the A and D forms of CBFb-MYH11/inv(16), AML1-ETO/t(8;21)(q22;q22), E2A-PBX-1/t(1;19)(q23;p13), MLL-AF4/t(4;11)(q12;q23) and TEL-AML1/ t(12;21)(p12;q22).INTERESTING FEATURES
Myelodysplastic syndrome can conceptualized as a phase in a myeloid neoplasm with evolution over time at genomic level with dynamic clinical presentation. Evolution can be to a myelodysplastic / myeloproliferative neoplasm, or acute myeloid leukemia. Components of genomic evolution include driver mutations; RAS is the best known but other genes may be involved as well. This patient started with myelodysplastic syndrome with remissions and relapses; progressed to chronic myelomonocytic leukemia after 7 years; and transformed to acute myeloid leukemia 4 months later.
Blast percentage, considered in isolation, does not dictate management decisions. In 09/2011, blast percentage was 7% in aspirate smears, 8% blasts in peripheral blood. However, peripheral blood absolute blast count was 3.6K/ul As such, the absolute blast count increased many times over, in spite of patient being on chemotherapy, while never meeting formal criteria of 20% blasts to diagnose acute myeloid leukemia. In our experience, when blast percentage is less than 20% in bone marrow and peripheral blood, pathologists are reluctant to diagnose acute myeloid leukemia even though absolute blast counts are elevated markedly. In such situations, some flexibility is necessary to enable patients to qualify for treatment protocols and get optimal treatment. In our opinion, blast count of 3.6K in peripheral blood would be enough to diagnose transformation to acute myeloid leukemia irrespective of blast percentage. The prognostic implications can be judged by the fact that this was a terminal event in this case. Towards the end, peripheral AMC increased from 10.4K to 41.3K; a four fold increase over 6 days; blast count went from 8 to 16K over 3 days.PROPOSED DIAGNOSIS
Myelodysplastic syndrome evolving to chronic myelomonocytic leukemia evolving to acute myeloid leukemia
CONSENSUS DIAGNOSIS
Acute myeloid leukemia with myelodysplasia-related changes, arising from chronic myelomonocytic leukemia
| Bone marrow clot 1 |  |
| Bone marrow clot 2 |  |
| Bone marrow aspirate 1 |  |
| Bone marrow aspirate 2 | 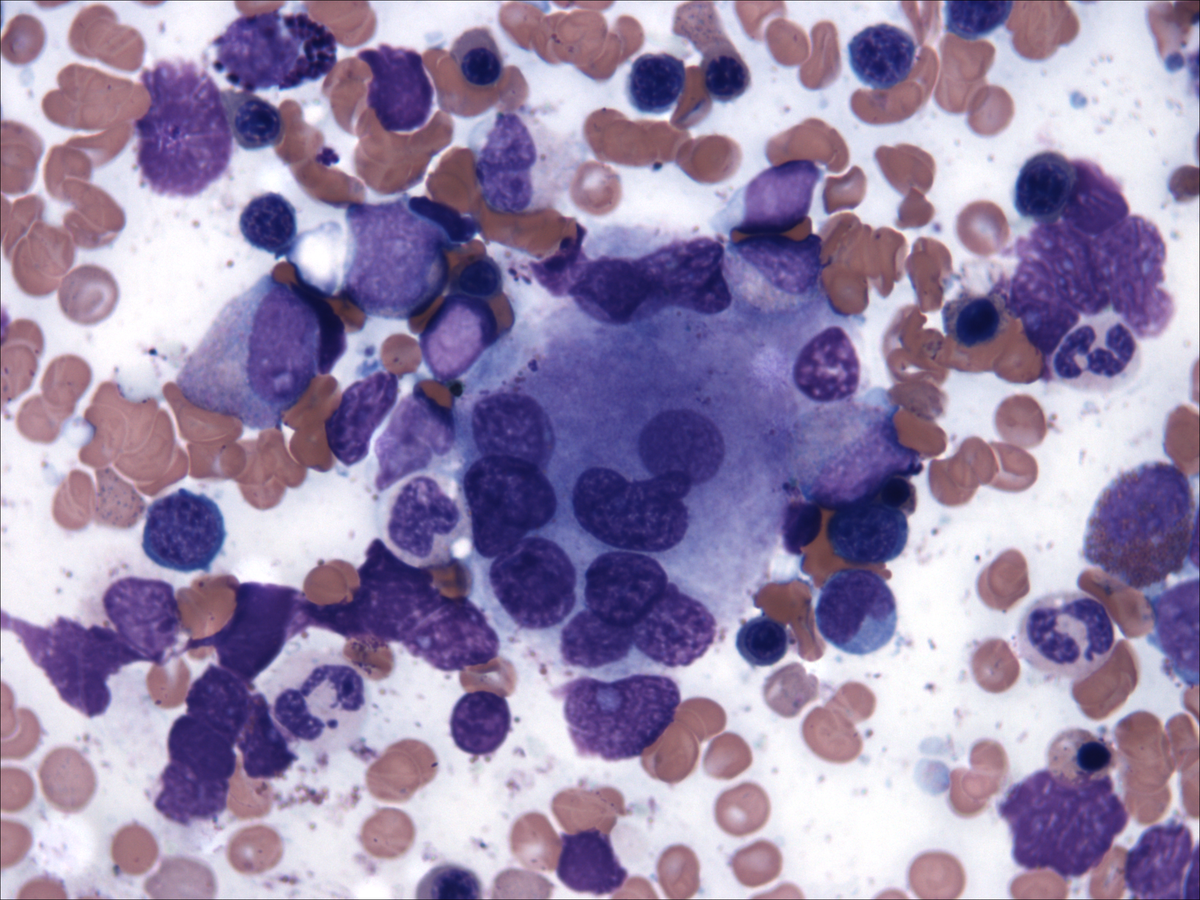 |
| Bone marrow aspirate: butyrate | 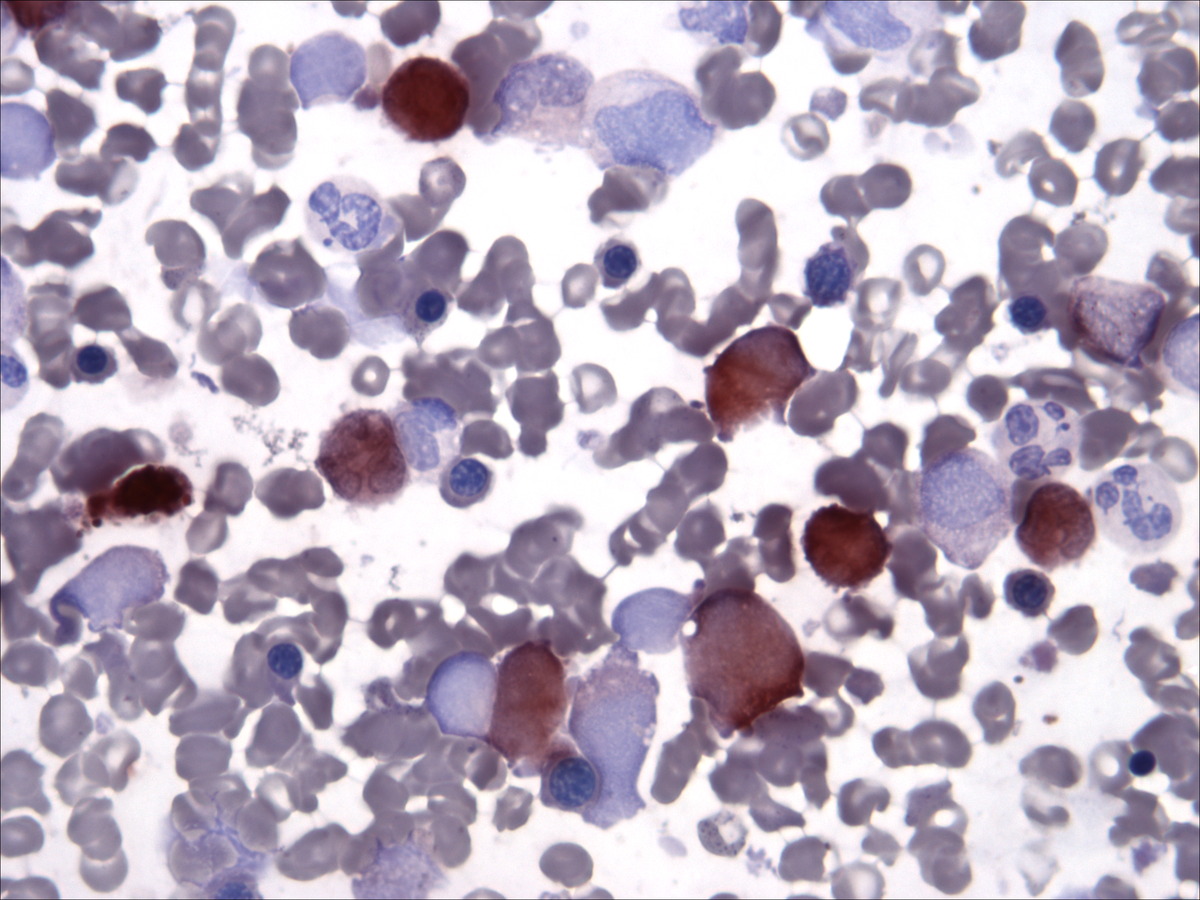 |
| Peripheral blood |  |